Background
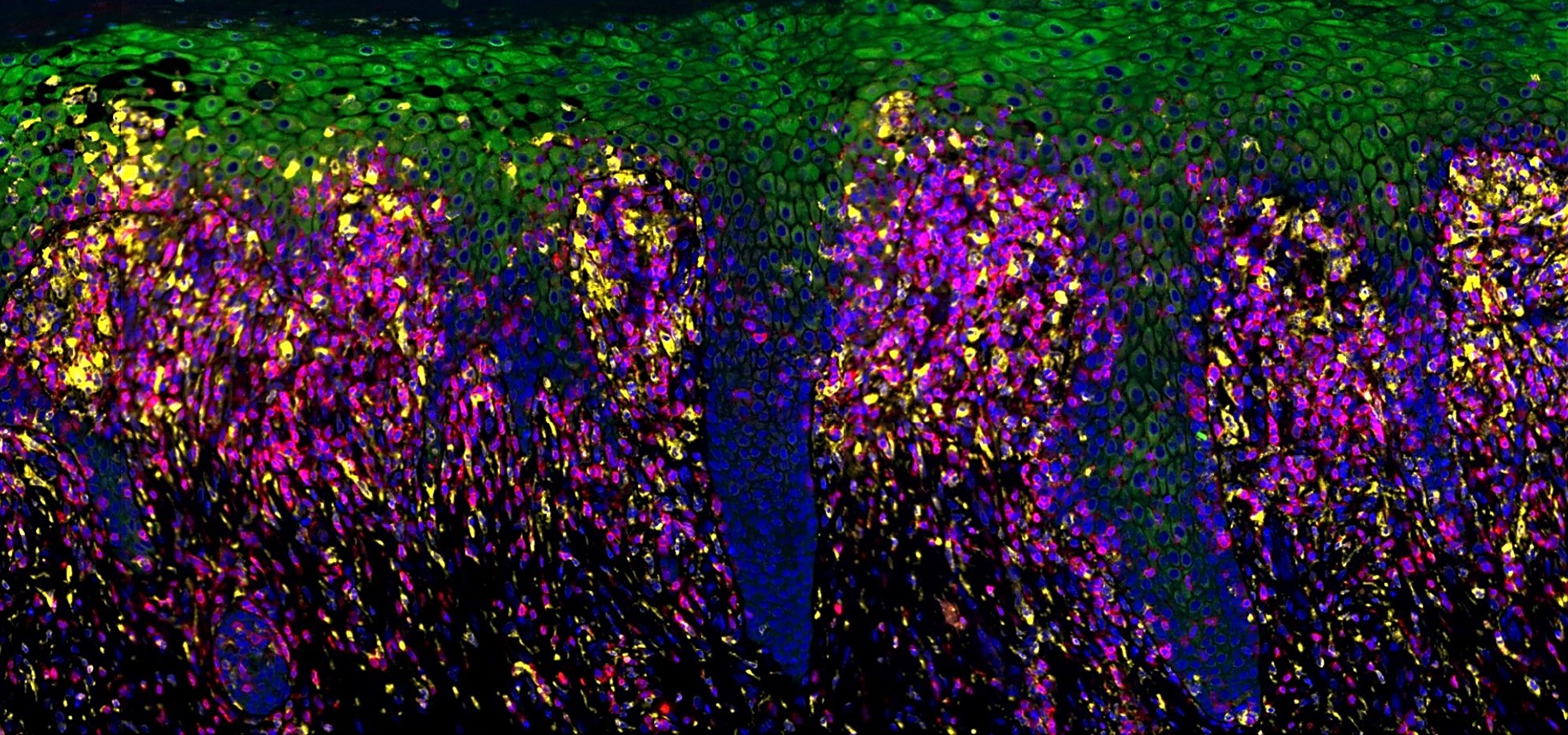
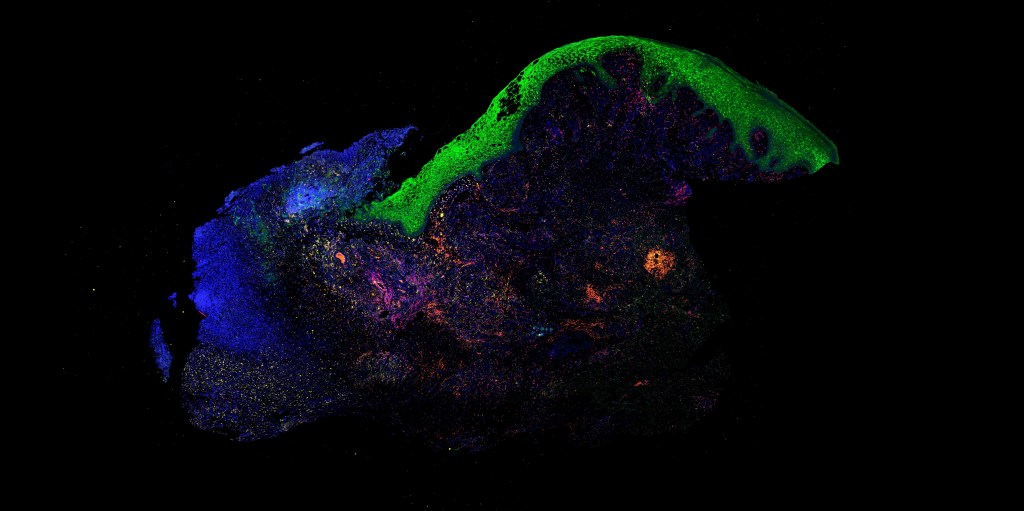
The majority of skin wounds progress uneventfully through the sequential phases of healing, with the magnitude of the inflammatory response linked to the extent of the resultant scar. However, chronic wounds, including venous leg ulcers, diabetic foot ulcers and pressure ulcers, are thought to become ‘stuck’ in a chronically inflamed state, unable to heal effectively. Multiple immune and stromal cells, including macrophages, neutrophils, lymphocytes and keratinocytes play important roles in physiological and, most likely, pathophysiological healing. Macrophages are phenotypically plastic cells implicated both in supporting the ‘normal’ repair process, assisting angiogenesis, lymphangiogenesis and collagen deposition, and in playing detrimental roles in aberrant healing scenarios, with studies indicating that macrophages are dysfunctional, exhibiting unrestrained activation in the context of impaired healing in mouse and human. However, we lack a full appreciation of the dynamics of monocyte-macrophage recruitment and phenotype in acute skin wounds and macrophage biology has never been studied in a genetically tractable mammalian model of chronic wounds. An improved understanding of the acute repair program and how it becomes deranged to generate chronic wounds is urgently needed to inform new wound therapeutics. Exploiting resolution pathways may provide novel therapeutic strategies to stem the growing patient and healthcare burden that healing disorders inflict, perhaps by triggering resolution of chronic inflammation in non-healing wounds or reducing fibrosis.
Research Overview
We are interested in the events that influence whether a skin wound will heal acutely or develop into a chronic non-healing wound. We are focusing on the role of macrophages in determining wound outcome by addressing questions such as ‘how and when do macrophages become dysfunctional in aberrant healing scenarios’.
We use a variety of complementary approaches, including multiphoton live imaging of skin repair in mouse, single cell transcriptomics, flow cytometry and immunohistochemical techniques. We also take advantage of in vitro assays and human wound biopsies to help dissect mechanism and provide translational relevance.
Our second main research direction involves exploring whether pro-resolving pathways can be harnessed to drive healing, including rescue of chronic wounds, in order to develop novel therapies to improve healing and rescue chronic wounds.

Current Projects
- Macrophage phenotypes and subsets in skin healing and chronic wounds
- Angiogenesis and lymphangiogenesis
- Investigating pro-resolving pathways in wound repair and repair failure
- Neutrophil heterogeneity and function in chronic wounds